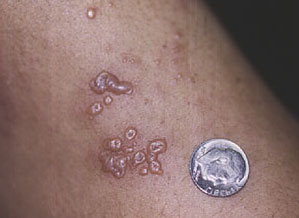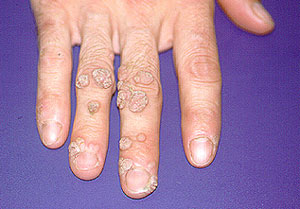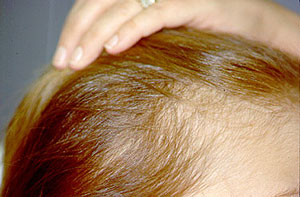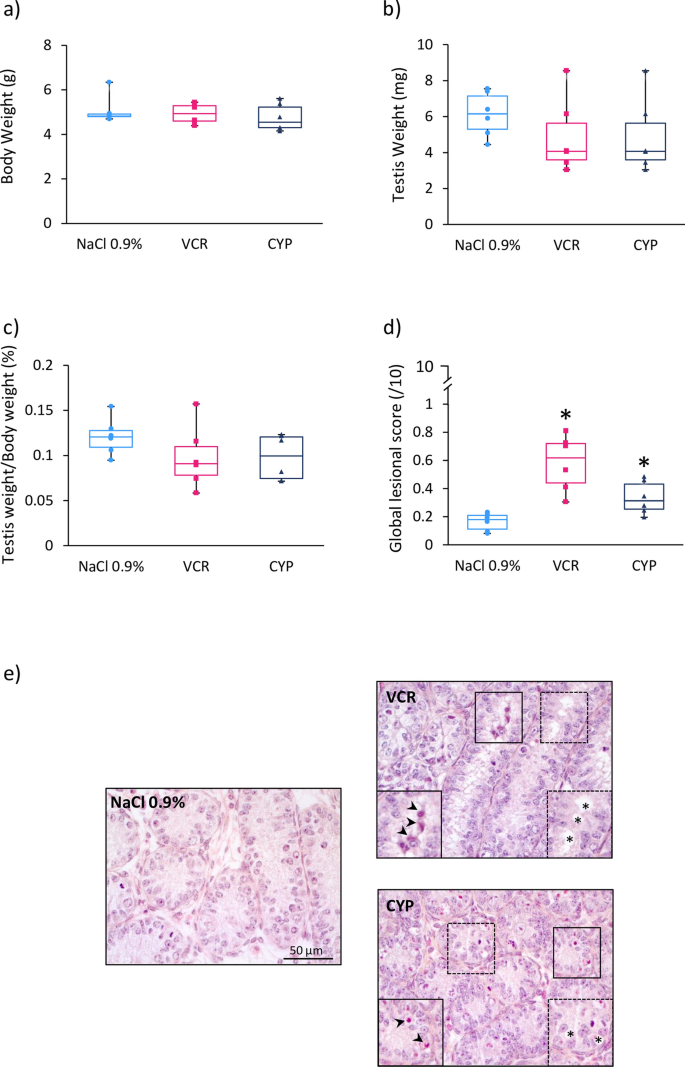
Paradoxical risk of reduced fertility after exposure of prepubertal mice to vincristine or cyclophosphamide at low gonadotoxic doses in humans | Scientific Reports
Cyclophosphamide: As bad as its reputation? Long-term single centre experience of cyclophosphamide side effects in the treatment

Rituximab versus intravenous cyclophosphamide in patients with connective tissue disease-associated interstitial lung disease in the UK (RECITAL): a double-blind, double-dummy, randomised, controlled, phase 2b trial - The Lancet Respiratory Medicine

Safety of Cyclophosphamide Therapy in Autoimmune Rheumatic Diseases Subramanian R, Pathak H, Ravindran V - Indian J Rheumatol

Chemotherapy and Biotherapy Agents. Objectives At the completion of this session the participant will be able to: ◦ Identify common side effects for individual. - ppt download

Long-Term Outcome After Cyclophosphamide Treatment in Children With Steroid-Dependent and Frequently Relapsing Minimal Change Nephrotic Syndrome - ScienceDirect
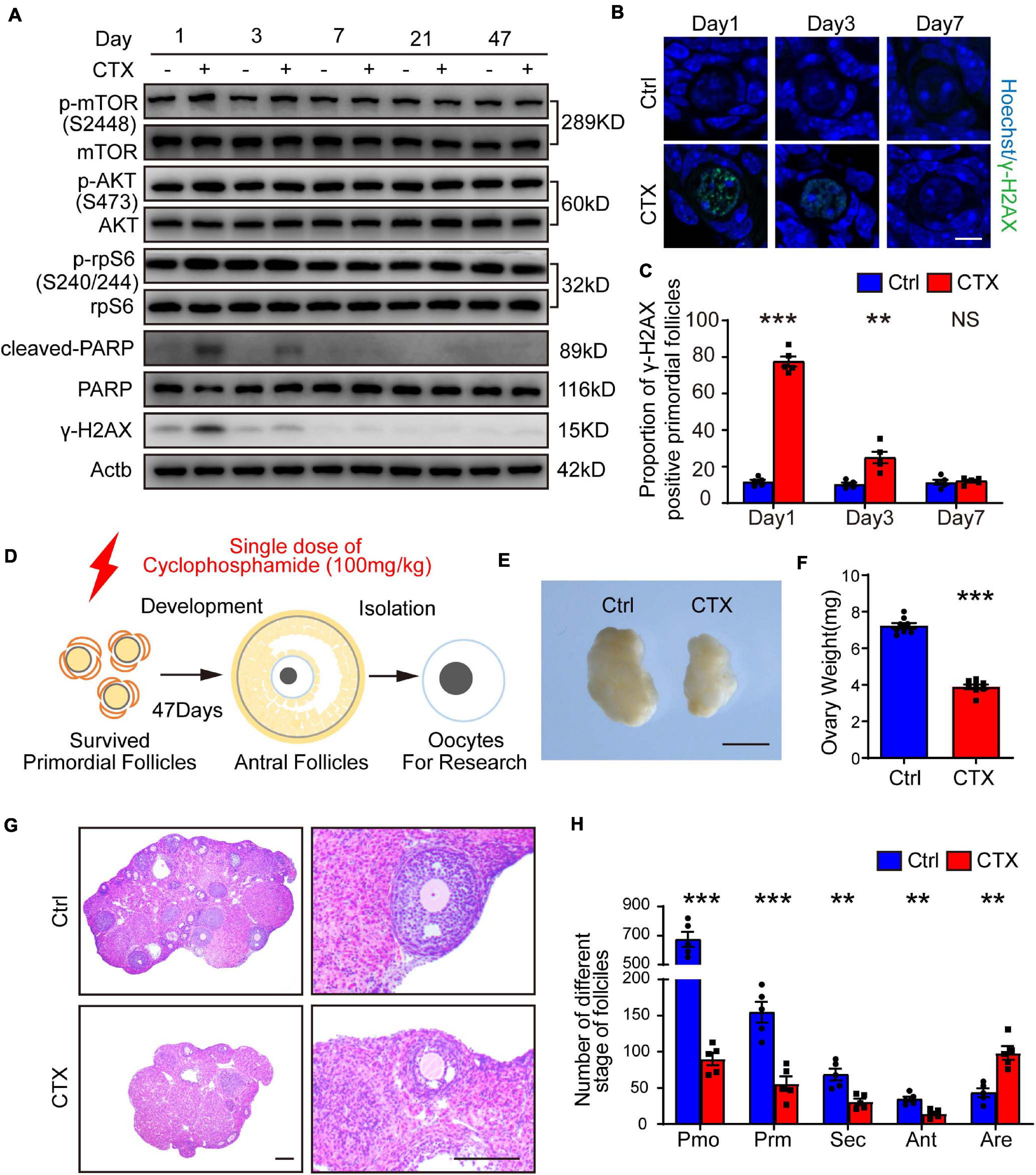
Frontiers | Cyclophosphamide Exposure Causes Long-Term Detrimental Effect of Oocytes Developmental Competence Through Affecting the Epigenetic Modification and Maternal Factors' Transcription During Oocyte Growth

Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial - The Lancet